PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES (PNAS)
Study by Sujay S. Kaushala, Gene E. Likensb, Michael L. Paced, Ryan M. Utze, Shahan Haqa, Julia Gormana, and Melissa Gresea
Freshwater Salinization Syndrome on a Continental Scale
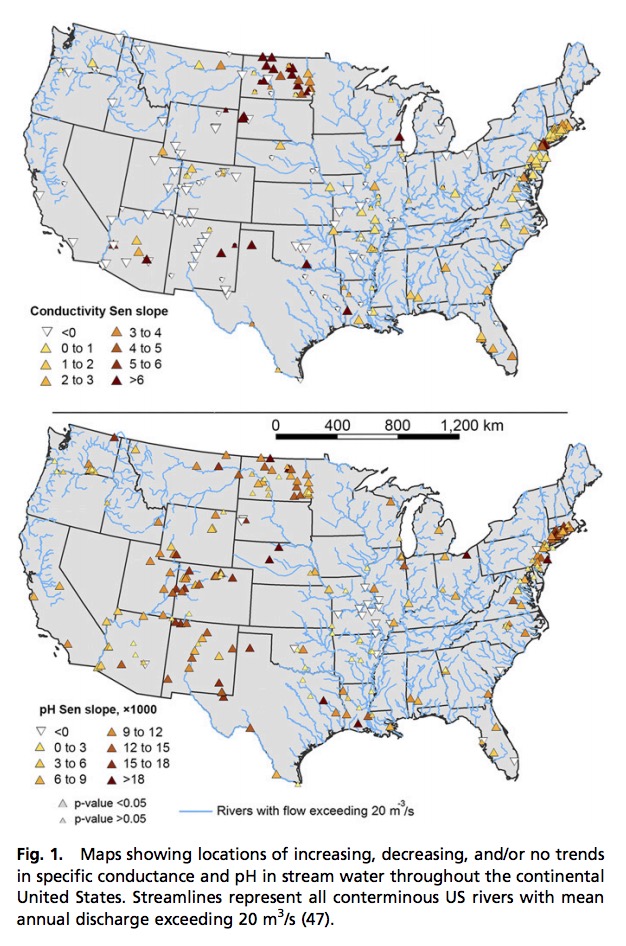 Salt pollution and human-accelerated weathering are shifting the chemical composition of major ions in fresh water and increasing salinization and alkalinization across North America. We propose a concept, the freshwater salinization syndrome, which links salinization and alkalinization processes. This syndrome manifests as concurrent trends in specific conductance, pH, alkalinity, and base cations. Although individual trends can vary in strength, changes in salinization and alkalinization have affected 37% and 90%, respectively, of the drainage area of the contiguous United States over the past century. Across 232 United States Geological Survey (USGS) monitoring sites, 66% of stream and river sites showed a statistical increase in pH, which often began decades before acid rain regulations. The syndrome is most prominent in the densely populated eastern and midwestern United States, where salinity and alkalinity have increased most rapidly. The syndrome is caused by salt pollution (e.g., road deicers, irrigation runoff, sewage, potash), accelerated weathering and soil cation exchange, mining and resource extraction, and the presence of easily weathered minerals used in agriculture (lime) and urbanization (concrete). Increasing salts with strong bases and carbonates elevate acid neutralizing capacity and pH, and increasing sodium from salt pollution eventually displaces base cations on soil exchange sites, which further increases pH and alkalinization. Symptoms of the syndrome can include: infrastructure corrosion, contaminant mobilization, and variations in coastal ocean acidification caused by increasingly alkaline river inputs. Unless regulated and managed, the freshwater salinization syndrome can have significant impacts on ecosystem services such as safe drinking water, contaminant retention, and biodiversity.
Salt pollution and human-accelerated weathering are shifting the chemical composition of major ions in fresh water and increasing salinization and alkalinization across North America. We propose a concept, the freshwater salinization syndrome, which links salinization and alkalinization processes. This syndrome manifests as concurrent trends in specific conductance, pH, alkalinity, and base cations. Although individual trends can vary in strength, changes in salinization and alkalinization have affected 37% and 90%, respectively, of the drainage area of the contiguous United States over the past century. Across 232 United States Geological Survey (USGS) monitoring sites, 66% of stream and river sites showed a statistical increase in pH, which often began decades before acid rain regulations. The syndrome is most prominent in the densely populated eastern and midwestern United States, where salinity and alkalinity have increased most rapidly. The syndrome is caused by salt pollution (e.g., road deicers, irrigation runoff, sewage, potash), accelerated weathering and soil cation exchange, mining and resource extraction, and the presence of easily weathered minerals used in agriculture (lime) and urbanization (concrete). Increasing salts with strong bases and carbonates elevate acid neutralizing capacity and pH, and increasing sodium from salt pollution eventually displaces base cations on soil exchange sites, which further increases pH and alkalinization. Symptoms of the syndrome can include: infrastructure corrosion, contaminant mobilization, and variations in coastal ocean acidification caused by increasingly alkaline river inputs. Unless regulated and managed, the freshwater salinization syndrome can have significant impacts on ecosystem services such as safe drinking water, contaminant retention, and biodiversity.
The abundance, integrity, and distribution of Earth’s fresh water are critical for human welfare. Only 2.5% of Earth’s water is fresh, and dissolved salts within this water determine the degree to which it can be used for drinking, industry, agriculture, and energy production (1, 2). The primary causes of freshwater salinization around the world are agriculture, resource extraction, and land clearing (3, 4). Relatively recently, human salt inputs are increasingly becoming recognized as important over large geographic scales (5, 6). Alkalinization has received relatively less recognition as a related environmental issue, but increases in alkalinity are also generated by salt pollution, accelerated weathering, and microbial processes that influence water chemistry (7). Salinization and alkalinization of fresh water can be interconnected processes that deteriorate water quality over a range of climates, but it is typically assumed that these processes are related only in arid regions (6–9). In particular, increasing concentrations of dissolved salts with strong bases and carbonates can increase the pH of fresh water over time, linking salinization to alkalinization.
We propose a concept, the freshwater salinization syndrome, which links salinization and alkalinization processes along hydrologic flow paths from small watersheds to coastal waters. The freshwater salinization syndrome manifests to varying degrees as concurrent trends in specific conductance, pH, alkalinity, and base cations (i.e., sodium, calcium, magnesium, and potassium). There are at least three primary categories of salt sources driving the freshwater salinization syndrome: (i) anthropogenic salt inputs (e.g., road salts, sewage, brines, sodic/saline irrigation runoff); (ii) accelerated weathering of natural geologic materials by strong acids (e.g., acid rain, fertilizers, and acid mine drainage); and (iii) human uses of easily weathered resource materials (e.g., concrete, lime), which cause increases in salts with strong bases and carbonates (6, 9–13). Although previous work has focused primarily on sodium chloride as a dominant form of salt pollution, increases in different mixtures of salt ions such as sodium, bicarbonate, magnesium, sulfate, etc., as part of the freshwater salinization syndrome produce differential toxicity to aquatic life (14, 15), and further supports the need for studying the dynamics of dissolved salts holistically. Although environmental impacts of the freshwater salinization syndrome are still poorly understood, symptoms can include: changes in biodiversity due to osmotic stress and desiccation, corrosion of infrastructure, increased contaminant mobilization, enhanced river carbonate transport, and impacts on coastal ocean acidification caused by increasingly alkaline river inputs.
 Over geologic time, salinization and alkalinization of water have naturally influenced the distribution and abundance of Earth’s life (16). However, rates of salinization and alkalinization in the Anthropocene are strongly influenced by three primary factors: atmospheric deposition, geology, and land use (7). Atmospheric acid deposition accelerates chemical weathering over large geographic scales due to chemical dissolution and ion exchange in rocks and soils (7, 12, 17). Accelerated weathering of geological substrates can contribute to increased concentrations of major ions in water including bicarbonate, calcium, magnesium, and potassium (7, 12, 18–21). Finally, land-use activities enhance the anthropogenic inputs of easily weathered materials, including agricultural lime and impervious surfaces made of concrete, which can impact chemistry of drainage waters (13, 22, 23). Road salt and brines from fracking fluids further contribute to ion exchange in soils and sediments, and can increase salinity and alkalinity via changes in base cations and bicarbonate concentrations (11). Mining and resource extraction contribute to accelerated weathering of rocks and soils and the release of major ions and salts (21, 24–26). Finally, wastewater, water reuse, and irrigation practices associated with urban and agricultural land use can increase salinity and alkalinity in fresh water (7, 10, 27). All of these factors synergistically contribute to the freshwater salinization syndrome. The directionality and severity of trends for different salt ions, alkalinity, and pH are influenced on a regional scale by dilution capacity of rivers due to water demand, changes in river runoff due to climate variability, and sea level rise and saltwater intrusion into freshwater sections of rivers and estuaries (6, 28, 29). A holistic understanding of concurrent patterns in salinization and alkalinization on a continental scale is important for identifying and prioritizing regional management needs, given that interactions between different salts, specific conductance, and pH can influence aquatic life, ecosystem functioning, and services (14, 15, 19, 30).
Over geologic time, salinization and alkalinization of water have naturally influenced the distribution and abundance of Earth’s life (16). However, rates of salinization and alkalinization in the Anthropocene are strongly influenced by three primary factors: atmospheric deposition, geology, and land use (7). Atmospheric acid deposition accelerates chemical weathering over large geographic scales due to chemical dissolution and ion exchange in rocks and soils (7, 12, 17). Accelerated weathering of geological substrates can contribute to increased concentrations of major ions in water including bicarbonate, calcium, magnesium, and potassium (7, 12, 18–21). Finally, land-use activities enhance the anthropogenic inputs of easily weathered materials, including agricultural lime and impervious surfaces made of concrete, which can impact chemistry of drainage waters (13, 22, 23). Road salt and brines from fracking fluids further contribute to ion exchange in soils and sediments, and can increase salinity and alkalinity via changes in base cations and bicarbonate concentrations (11). Mining and resource extraction contribute to accelerated weathering of rocks and soils and the release of major ions and salts (21, 24–26). Finally, wastewater, water reuse, and irrigation practices associated with urban and agricultural land use can increase salinity and alkalinity in fresh water (7, 10, 27). All of these factors synergistically contribute to the freshwater salinization syndrome. The directionality and severity of trends for different salt ions, alkalinity, and pH are influenced on a regional scale by dilution capacity of rivers due to water demand, changes in river runoff due to climate variability, and sea level rise and saltwater intrusion into freshwater sections of rivers and estuaries (6, 28, 29). A holistic understanding of concurrent patterns in salinization and alkalinization on a continental scale is important for identifying and prioritizing regional management needs, given that interactions between different salts, specific conductance, and pH can influence aquatic life, ecosystem functioning, and services (14, 15, 19, 30).
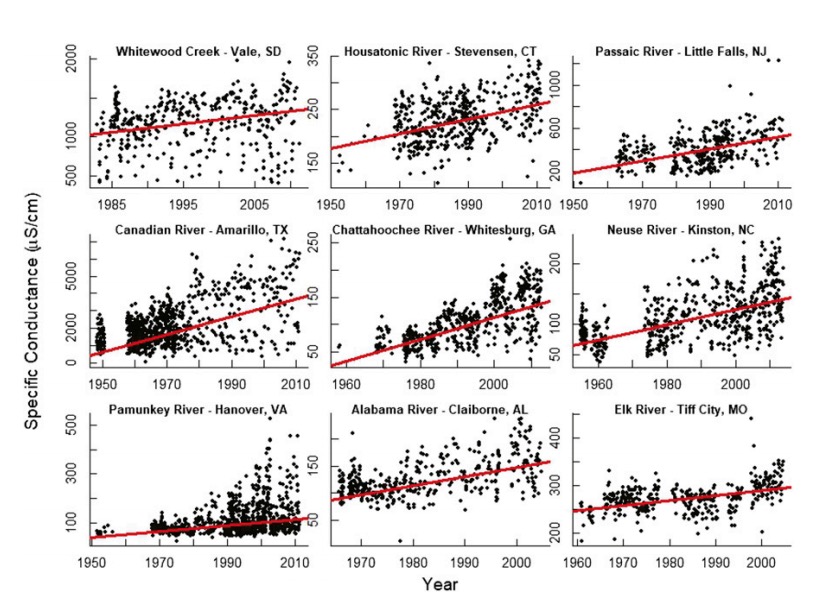
In this study, we (i) quantify concurrent trends in salinization rates, alkalinization rates, and base cation concentrations in fresh water on a continental scale and (ii) present a conceptual framework, called the freshwater salinization syndrome, which illustrates and explains how coupled salinization and alkalinization can occur. We quantified long-term trends in specific conductance, base cations, alkalinity, and pH in 232 stream and river sites sampled by the USGS throughout North America. Although these sites include both streams and rivers, we will mainly refer to them as rivers throughout. Watersheds drained areas primarily in the United States, but also extended into Canada, and spanned diverse regions and climates. Watersheds also sometimes drained major reservoirs. Surprisingly, we found that increasing trends in the pH of rivers occurred decades before US Clean Air Act Amendments in 1990, which specifically targeted reductions in acid rain (17). After 1990, a decrease in acidic precipitation and acid mine drainage may be expected to contribute to an increase in alkalinity and pH in the latter part of the twentieth century, but such reductions in acidification would not adequately explain earlier concurrent increases in specific conductance, base cations, alkalinity, and pH beginning in the early and middle twentieth century (31). The freshwater salinization syndrome illustrates how anthropogenic salts and humanaccelerated weathering of natural and anthropogenic substrates synergistically drive salinization and alkalinization due to a plethora of salts containing strong bases and carbonates with potential for acid neutralization. In addition, increased sodium from salt pollution displaces other base cations on soil exchange sites, which further increases pH and contributes to alkalinization. From a biological perspective, in-stream microbial processes such as denitrification, sulfate reduction, etc., can further increase pH due to biochemical reactions that generate alkalinity. Finally, photosynthesis and gross primary production in eutrophic fresh waters in the Anthropocene can raise pH through depletion of total dissolved carbonate relative to equilibrium with CO2. The freshwater salinization syndrome can impact ecosystem services related to drinking water quality (6, 7, 9), economic costs related to switching water treatment and sources (9, 10), impacts on aquatic biodiversity (8), pipe corrosion and leaching of metals (7, 9), and shifting rates of coastal ocean acidification (32, 33).
…
Environmental Implications of the Freshwater Salinization Syndrome
The freshwater salinization syndrome currently poses direct and indirect concerns for ecosystem services and may influence rates of coastal ocean acidification. Elevated pH can contribute to ammonia toxicity and mortality for aquatic organisms (49, 60). Different salts influence toxicity to aquatic organisms leading to recent calls for regulating salt composition and concentrations in fresh water by government agencies (8). Elevated pH and base cations, such as calcium and magnesium, may reduce the bioavailability and toxicity of trace metals (60), but salinization may also enhance ion exchange and mobilization of trace metals from soils to streams (61). Increased sodium concentrations decrease soil aggregate stability and soil fertility through leaching of calcium and magnesium (62). Salinization from road salts can increase leaching and mobilization of carbon, nitrogen, and phosphorus from soils and sediments to streams (63). However, detailed work has also shown that alkalinization or rather sodium absorption ratio or exchangeable sodium percentage is responsible for increasing dissolved organic carbon and not salinization (64). Salinization also may directly influence the quality of different fractions of organic matter released from soils to streams (65). Elevated alkalinity can also stimulate production of nitrate by microbial nitrification, and salinization can alter the urban evolution of ecosystems and aquatic communities (22). Increased temperatures in rivers throughout the United States (66) may further influence weathering and other geochemical reactions, and interact with impacts of freshwater salinization.
Furthermore, increased pH, alkalinity, and salinity influence the amount and quality of inorganic and organic carbon transported by rivers, which can influence aquatic species and rates of coastal ocean acidification (32, 33, 56, 57). Changes in the pH and alkalinization of rivers also influence rates of CO2 evasion, which are important for global carbon budgets (67). From a coastal perspective, increased alkalinity can contribute to increased shell thickness of calcareous organisms such as found in zebra mussels in the Hudson River estuary (57), where there has been an increase in alkalinity over the past decade (Fig. 8). Estuarine waters of the Chesapeake Bay also have long-term positive trends in pH and alkalinity near tributaries (Fig. 8) (56). Overall, the effects of the freshwater salinization syndrome on the riverine carbon cycle and variations in coastal ocean acidification due to riverine inputs warrant further investigation (12, 32, 33).
The freshwater salinization syndrome can increase risks to the safety of drinking water and infrastructure. Elevated salt levels in drinking water can contribute to hypertension in people on sodium-restricted diets and is of concern to people requiring kidney dialysis (9). Salinization and alkalinization influence the corrosivity of water, and this can affect leaching of metals from pipes carrying drinking water (9, 68). Salinization increases corrosion of transportation infrastructure with United States economic costs estimated in the billions of dollars (69). Given increasing impacts on ecosystems and human welfare, increased salinization and alkalinization of fresh water is now a pervasive water quality issue, which may require aggressive management in both arid and humid climates across latitudes.
Download full version (PDF): Freshwater salinization syndrome on a continental scale
About the Proceedings of the National Academy of Sciences (PNAS)
www.nasonline.org/publications/pnas
One of the world’s most-cited multidisciplinary scientific serials, the Proceedings of the National Academy of Sciences publishes cutting-edge research reports, commentaries, reviews, perspectives, and colloquium papers spanning the biological, physical, and social sciences. PNAS is published weekly in print and daily online. PNAS Online contains the full text of all articles; access to the current content is available by subscription, but free access is available immediately to selected content as well as to the complete content of all issues six months after their publication in print.
Tags: Alkalinization, Freshwater, National Academy of Sciences, PNAS, Proceedings of the National Academy of Sciences, Runoff, Salinization






 RSS Feed
RSS Feed